771
Views & Citations10
Likes & Shares
The article presents experimental studies of the detection of aspartate
transaminase activity in the hepatic tissues of goats of Khizi-Khachmaz zone of
Azerbaijan in different seasons of the year for the period from December 2017
to January 2018, depending on the degree of invasion by parasites of animals.
Determination of the enzymatic activity was carried out
spectrophotometrically using a Folin reagent on a Specol 1500 spectrophotometer
(Analitik Jena).
The maximum peak of intensity of aspartate-transaminase enzymatic
activity of liver tissues of goats was revealed. The maximum value of the
enzyme activity helminthes in liver tissues was reached in Spring season in
March equal to 191.1 U/l of tyrosine per gram of tissue weight of the liver, and the minimum
was in December reaching 72.1 U/l of tyrosine per gram of wet weight of the
hepatic tissue.
Thus, experimental studies were conducted to identify
aspartate-transaminase activity of hepatic tissue in goats infested by
parasites in different seasons of year. Proceeding from the obtained data, it
can be stated that the season of the year and the climatic conditions of their
maintenance in farms significantly influences the aspartate-transaminase
enzymatic activity of goat homogenates.
Thus, comparing the average values of aspartate-transaminase activity in
homogenates of non-modal tissues isolated from goat liver in different seasons
of the year, it should be noted that their difference is significant (P>0.96).
The maximal intensity peak of aspartate-transaminase activity in liver
tissues of goats infested by parasites was found in winter (186.4 μg) and
spring (168.6 μg) periods of the year and the minimum was found in summer
season equal to 72.1 μg of tyrosine per gram of wet weight of helminth.
Keywords: Enzyme activity,
Aspartate transaminase, Goats, Parasites
INTRODUCTION
The biology of pathogens of
helminthes transmitted to humans through meat and meat products, in conjunction
with the climatic and socio-economic characteristics of various administrative
ethnic regions of the country, determine the peculiarity of the epidemiology
and epizootology of these invasions in each specific area and zoogeographical
zone. The fight against these biohelminthiasis is based on a comprehensively
differentiated approach to each nosoform; it requires a differentiated,
integrated, dynamic and flexible approach to the problems under consideration,
the successful solution of which depends on the timeliness and regularity of
the activities. Nematodes are pathogenic parasites causing disease in the host.
They usually live in the host's digestive system. It causes weight loss in
sheep and goats, feeding on the blood of the host and also causes anemia [1].
Proteolytic enzymes play an
important role in the study of nutrition of some nematodes and mainly in the
study of the nutrition of tapeworms [2].
One of the important factors
determining the extent of spread and intensity of invasions is the time of year
and the climatic conditions in the farms [3].
It is noted that the increase in
the physiological activity of parasites occurs in spring, in summer and to a lesser extent
in autumn. All this is due to the biological cycle of helminthes in the host
and in the environment, the nature of animal nutrition, phenomena of latent
invasion and an increase or suppression of the sexual activity of helminthes in
the host organism [4].
In addition, insufficient
veterinary care contributes to the development and transmission of nematodes.
This problem is especially evident in small ruminants, sheep and goats.
Consequences of nematode invasion include: reduced feed intake, reduced
immunity, reduced fertility, reduced milk production, treatment costs and death
in critical infections [5].
At trematodoses in varying
degrees, a decrease in the quality and nutritional value of meat is recorded,
especially in protein, which is accompanied by a decrease in calories by
6.7-21.9% [6,7].
Parasites can have a twofold
effect on the host [8]. On the one hand, they stimulate the immune response,
resulting in a number of cellular and humoral response phenomena and on the
other hand, they cause inhibition of the functional and proliferative activity
of lymphoid tissue cells, leading to the development of secondary immune
deficiencies. This contributes to a dramatic change in the nature of the
relationship in the host-parasite system and helps the survival of the latter in
the host organism [9,10].
The aim of our research was
to study the dynamics of aspartate transaminase enzymatic activity of goat
liver tissues taken from slaughtered goats of the Khiza - Khachmaz zone of
Azerbaijan in different seasons of the year.
MATERIALS AND RESEARCH METHODS
The object of the study was
goats (infected and uninfected) from the regions of Azerbaijan (Khizi and
Khachmaz) for the period from December 2017 to December 2018 inclusive. Totally
65 goats were investigated. For the experiments, 8-9 month old animals were
taken.
The material for the study
was the liver of goats (65), cut in winter (December, January, February),
spring (March, April, May), summer (June, July, August) and autumn (September,
October, November) periods of the year.
Experimental studies to
determine the enzymatic activity were performed spectrophotometrically using
Folin's reagent on a Specol 1500 spectrophotometer (Analitik Jena).
Per unit of enzymatic
activity, the amount of enzyme catalyzing in 30 minutes hydrolysis of 1 g of
protein not precipitated with trichloroacetic acid was taken. In this case, 1 g
was 25% of the protein taken for the enzymatic reaction.
RESEARCH RESULTS AND DISCUSSION
In winter, spring, autumn
and summer, helminthes were removed from the liver of slaughtered goats,
thoroughly washed with 0.9% sodium chloride solution, then dried with filter
paper, followed by grinding and homogenizing with three volumes of 0.025 N HCl
at room temperature. In this case, the homogenizer was placed in a vessel with ice.
Casein was used as a substrate.
The enzymatic activity of
goat liver tissues was determined by the method of Kunitz and Anson modified by
Orekhovich [9].
1 ml of the helminthes
homogenate was added to a solution of 1 ml of casein. The mixture was incubated
for 1 hour in a thermostat at 37°C, then 3 ml of a 5% solution of
trichloroacetic acid was added. Samples were left for 1 h to form a
precipitate, followed by centrifugation. Next, 1 ml of centrifugate was taken 2
ml of 0.5 M NaOH and 0.9 ml of Folin solution was added. Pre-folin solution was
diluted three times with distilled water. The prepared samples were left for 10
min before the development of a stable color.
Measurements of the optical
density of the samples were carried out on a Specol 1500 spectrophotometer
(Analitik Jena) at a wavelength of 750 nm. Samples in which trichloroacetic
acid was added together with the filtrate served as controls. Enzyme activity
was expressed in μg of tyrosine in 1 ml. The results were recalculated for 1 g
of the wet weight of the worms.
Quantitative data on the
determination of enzyme activity in the hepatic tissue of goats infested by
parasites was carried out in winter, autumn, spring and summer seasons of the
year (Table 1).
The maximal intensity peak of aspartate transaminase
activity in helminthes in liver tissues of goats invaded by parasites was found
in spring (191.1 U/l) and summer (168.6 U/l) periods and the minimum in winter
season was 72.1 U/l.
In invasive animals, an
increase in aspartate transaminase activity in liver helminthes tissues is
observed, reaching a maximum value in the spring, which is a result of
intoxication of the animal. However, in December, there was a decrease in
activity (72.1 U/l) and in January an increase in enzymatic activity to 88.3
U/l was observed, followed by a decrease in the level of enzymatic activity in
February to 82.2 U/l.
Comparing the average values
of aspartate-transaminase activity in homogenates of the tissues of the liver
of goats, invaded by helminthes, in different seasons of the year, it should be
noted that their difference is significant (P>0.96).
In conclusion, it should be noted that the maximum peak of enzymatic activity
in goat liver tissues reaches in winter season and spring season of the year
and is characterized by the highest rates in March and June and the lowest in
December, reaching 72.1, 88.3 and 82.2 U/l, respectively.
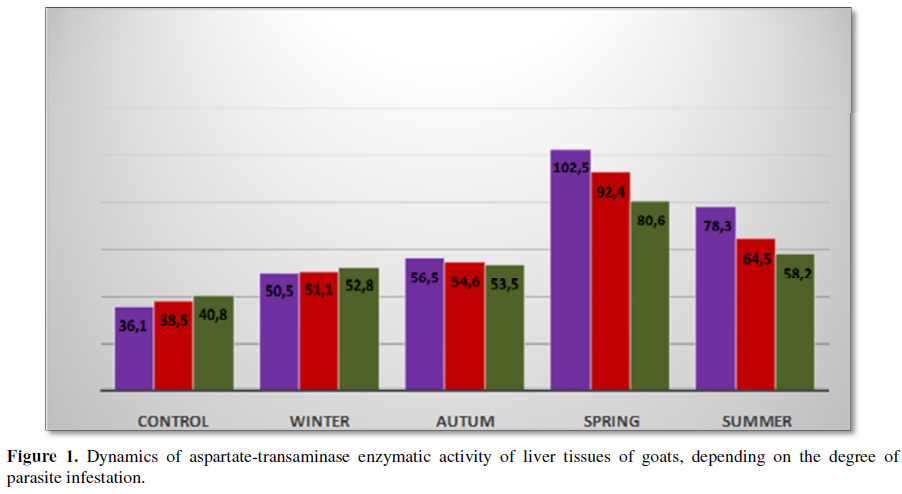
A different picture is observed in the tissues of the
liver of goats infested by parasites (Figure 1 and Table 2).
Thus, comparing the average values of
aspartate transaminase activity in homogenates of non-mode tissues isolated
from goat liver in different seasons of the year, it should be noted that their
difference is significant (P>0.96). In conclusion, it should be noted that the maximum peak of
enzymatic activity in the tissues of goat liver reaches in the spring season
and summer season of the year and is characterized by the highest rates in
March, and the lowest in December reaching 102.5 U/L and 50.5 U/L,
respectively.
FINDINGS
1.
Experimental studies were carried out to identify the aspartate
transaminase activity of the liver tissue in goats infested by parasites in
different seasons of the year. Based on the data obtained, it can be stated
that the aspartate transaminase enzymatic activity of goat liver homogenates is
significantly influenced by the season of the year and the climatic conditions
of their keeping in farms.
2.
The maximum peak intensity of aspartate transaminase activity in the
tissues of helminthes of goats liver infested by parasites in spring (191 U/L)
and summer (168 U/L) periods of the year and the minimum in winter season
reaching 72 U/L in December.
3.
The maximum peak intensity of aspartate transaminase activity in the
tissues of goat liver infested with parasites was found in the spring (102.5
U/L) and summer (78.3 U/L) periods of the year, and the minimum in the winter
season reaching 50.5 U/L in December.
1. Hale
M (2006) Managing internal parasites in sheep and goats. Available at: http://www.attra.ncat.org/attra-pub/PDF/parasitesheep.pdf
1-800-346-9140
2. Dubovskaya
AY (1973) Study of proteolytic activity in some species of cestodes. J
Parasitol 2: 154-158.
3. Akbaev
M Sh, Vasilevich FI (1992) Parasitology and invasive animal diseases. M.
Agropromizdat.
4. Abdullaev
VM, Gudkova A, Sorokina IB (2000) The dynamics of pathogenicity of bacteria in
the gastrointestinal tract of cattle with helminthiases. Innovative methods in
veterinary medicine. Mat. International Scientific Conference. FGOU VPO Ivanovo
State Agricultural Academy, Ivanovo 2: 99-101.
5. Fikru
R, Sori T, Dhuguma R, Kiros Y (2006) Epidemiology of gastrointestinal parasites
of ruminants in Western Oromia, Ethiopia. Int J Appl Res Vet Med 4: 51-57.
6. Menkir
MS, Uggla A, Waller PJ (2006) Epidemiology and seasonal dynamics of
gastrointestinal nematode infections of sheep in a semi-arid region of eastern
Ethiopia. Vet Parasitol 143: 311-321.
7. Muromtsev
AB (2008) The main helminthes infections of ruminants in the Kaliningrad
region: Author. diss. Dr. veterinary. Sciences: 03.00.19/Muromtcev Aleksandr
Borisovich. - St. Petersburg, b. 41 sec. 13.
8. Saveliev
AA (2005) Metabolic processes and meat quality in animals that are
spontaneously infected by trematodes and in the background of deworming.
Proceedings of the All-Russian Institute of Helminthology: Moscow, T. 41, pp:
312-317.
9. Kazharov
AZ (2013) Biohelminthiasis (echinococcosis, fascioliasis) of cattle of
different genotypes in the Kabardino-Balkarian Republic and the quality and
safety of meat products: author. Diss. PhD in Biological Sciences:
03.02.11/Kazharov Alim Zabitovich - M, p: 24.
10. Kryazhev
AL (2012) Cattle helminthofauna in the Vologda region. Curr Issues Vet Biol 4:
p: 28-32.